We bring patients and research together
- 42,000+ randomizations
- 60+ countries
- 12,500+ sites
Enterprise Trial Solutions
- Save up to 50% recruitment time and budget, with 3 unique solutions built to supercharge your clinical pipeline
How to increase clinical trial diversity
- Understanding the practical and psychological barriers to diverse patient participation
The A-Z of clinical trial branding from our Creative Director
Randomizations delivered with a proven track record
Patients were screened last year alone
Years of experience in clinical trial patient recruitment
Countries we have randomized patients in
We are celebrating 20 years of accelerating research
We’re dedicated to improving patients’ lives, by accelerating the development of new medical therapies. A proven track record of delivering over 42,000 randomizations, worldwide.
Testimonials
What our satisfied clients say about us
Contact us
Get in touch to find out how we can accelerate your clinical trials
patient recruitment
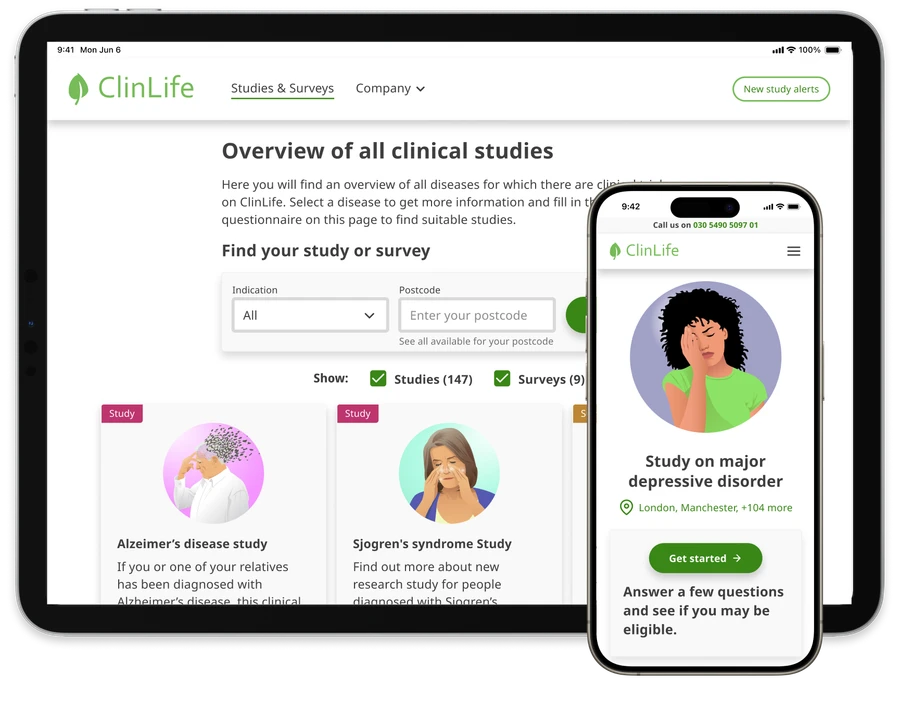
Enroll the patients you need
Having supported 2,500+ global studies, across 400 indications, we’re confident that we can expedite your patient recruitment and enrollment to your satisfaction, with minimal financial risk to you.
Willingness from black participants
Willingness from LGBTQ participants
Willingness from female participants
Willingness from female participants

study feasibility
Patient insights to ensure you avoid costly study delays and amendments
Use a data-driven approach to develop your protocol and choose the right regions and sites.
We recently conducted the largest international survey on diversity in the pharma industry, uncovering key drivers and barriers to clinical trial participation.
study creatives
Help your sites unlock their full potential with our study creatives
We equip your sites with the materials to recruit to their maximum potential.



Latest insights
Blog
readivo test
Blog
World Mental Health Awareness Month: How to recruit for mental health trials successfully
Blog
International Clinical Trials Day: Breaking down the barriers to trial participation through partnerships
Contact us
Get in touch to find out how we can accelerate your clinical trials
Speak to one of our experts today for personalized solutions and support